The Pemoline Molecule
Pemoline, first synthesized in 19131, is a CNS stimulant of the lesser known chemical family of oxazolines. The only other widely known member of this family is 4-Methylaminorex (a.k.a. 4-MAR, or "U4Euh"). Other than chemically, Pemoline differs from other stimulants in that it has very few symphatomimetic side effects, such as dry mouth, cardiac stress, or dilated pupils. It does not posess the jitteriness of caffeine either. The exact mechanism or site of action, via which pemoline acts, is not known with certainity, many studies indicate that it acts through dopaminergic mechanisms.
Clinically, in treating ADHD, Pemoline is normally given in dosages ranging between 37.5 and 75mg daily, with the maximum reccommended daily dose being 112.5 mg. Peak serum levels after single doses are reached within 2 to 4 hours and the serum half-life is approximately 12 hours. As Pemoline is insoluble in water, the powder is essentially tasteless. Pemoline is about five times as strong a CNS stimulant as caffeine, slightly stronger than methylphenidate (Ritalin) and but is not as strong as methamphetamine2. Pemoline does not suppress appetite like most other stimulants2. The LD50 of both Pemoline and its magnesium complex is 550 mg/kg in rats6. The melting point of Pemoline is 256-257°C, and is soluble to an extent of 50 µg/ml in water, the figures for Pemoline Magnesium being >300°C and 70 µg/ml respectively. Pemoline is also practically insoluble in ether and acetone. Slightly soluble in propylene glycol (1%) and in hot ethanol.
Pemoline is metabolized to an extent of 57% - the rest is excreted unchanged, primarily via the kidneys. Approximately 75% of an oral dose is recovered in the urine within 24 hours. Metabolites include pemoline conjugates, pemoline dione and mandelic acid. In studies conducted on primates, pemoline fails to demonstrate a potential for self-administration. However, this does not present proof for pemoline being non-addictive, only that the safety is relatively high.
Pemoline Magnesium, originally thought to be a chelate between pemoline and magnesium, displays certain pharmacological advantages over pemoline. The compound is the monohydrate of a equimolar mixture between pemoline and magnesium hydroxide. The absorption of pemoline magnesium is more rapid and the duration of action is extended, and the effect is more reliable5,6.
Experimental
Mandelic Acid Esterification
15.2 g (0.1 mole) DL-mandelic acid is dissolved in 250ml ethanol (at least 95%, but preferably absolute) containing 2ml conc. sulfuric acid. The mixture is held at reflux temperature for 3 hours and is then cooled to room temperature and allowed to stand overnight. 10g of Na2CO3 is added to neutralize the acid (some foaming may occur). The solution is filtered and the excess solvent is removed under vacuum. The residue is mixed with 50ml water and extracted with 3x50ml CH2Cl2. The water phase is discarded and the combined extracts are mixed and powdered sodium sulfate is added to the solution until it is free of any turbidity. The solution is suction filtered and the solvent is removed under vacuum. The residue is an almost colorless liquid, weighing around 12g (70%), pure enough for use in the next step. The ethyl mandelate can be further purified by vacuum distillation. The boiling point at atmospherical pressure is 253-255°C (103-105°C/2mmHg).
The preparation method for methyl mandelate is essentially the same. After removal of the solvent in the last step, methyl mandelate spontaneously forms a slightly off-white crystalline mass, pure enough for use in the next step. Can be purified by recrystallization from hot methanol, mp 54-56°C (bp 135°C at 12mmHg).
Esterification of Mandelic Acid Using an Iron(II)Sulfate Catalyst12
Place a mixture of mandelic acid (10mmol), alcohol (20mL) and Fe2(SO4)3·H2O (0.1g) in a 50mL two-necked flask , equipped with a condenser and mechanical stirrer. Reflux the mixture with stirring for 4 h, then remove the excess of alcohol by distillation under reduced pressure. Dilute the residue with ether (40mL) and filter off the catalyst. Wash the filtrate with 5% sodium bicarbonate solution and saturated brine solution. Dry over anhydrous magnesium sulfate, filter and evaporate to give the crude product, which is distilled. The pure product is isolated by recrystallization from petroleum ether.
Methyl ester, mp 53-54°C, ethyl ester mp 35-36°C. Yields 83% of theory.
Synthesis of Pemoline
Using Sodium Metal as the Base: Slowly add 2.3 grams (0.1 mole) of sodium metal cut into small pieces to 50ml MeOH. Let the exothermic reaction subside and cool the beaker with ice/water. Add 12.2 grams (0.1 mole) of guanidine nitrate, and let stir for 30mins. Add 0.1 mole mandelic ester (18 g ethyl mandelate or 16.6 grams methyl mandelate) dissolved in 50ml MeOH. Reflux the resulting solution until the evolution of NH3 ceases (about 60 min). Let cool, and acidify to pH 2-4 with conc HCl. Filter off the precipitated crystals, and rinse with cold water and MeOH. Air dry to give the title compound as fine white crystals, mp 256-257°C.
Using Sodium Hydroxide as the Base: A solution of guanidine freebase (Prepared by mixing a solution of 6.1g (0.05 mol) of Guanidine Nitrate in 30 ml of hot ethanol and a solution of 2g (0.05 mol) NaOH in 25 ml hot ethanol). 4.5g (0.025 mol) of ethyl mandelate in 5ml of ethanol is added to this boiling mix, and it is left at reflux for 30 minutes. The solution is cooled to room temperature, and 50 ml of cold distilled water is added. The solution is filtered and the precipitate is washed with cold water and MeOH in small portions. The precipitated pemoline is air dried on filter paper. Yield 90%, mp 256-257°C (dec).
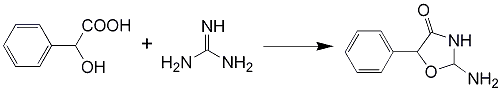
The Guanidine can also be substituted with an equimolar amount of Cyanamide, giving a yield of roughly 80-90%9,10.
Pemoline Magnesium6
To of Pemoline (4.4 g. 25 mmol) was added 50 ml of ethanol and 6 ml of 20 NaOH solution with rapid stirring. Following complete dissolution of the pemoline the solution was diluted with 100 ml distilled water. A solution of MgSO4·7 H2O (6.2g, 25 mmol) in 20 ml water was slowly added over a period of 5 min., followed by 5.5 ml of 20% NaOH solution to return the pH to 11. The resulting mixture was stirred for one hour, allowed to settle overnight, the clear aqueous solution decanted and the product obtained by filtration. The product was washed with water until the washings were neutral to litmus, then with a small quantity of acetone, followed by air drying.
Yield 5.9 g or 90% of theoretical.