For a number of years there has been extensive distribution and misuse of the clinical anaesthetic phencyclidine, 1-(1-phenylcyclohexyl)piperidine (PCP)1. This drug has been promoted and sold under various names such as "Crystal" and "Angel Dust" as well as misrepresented as being "Mescaline" or "Synthetic THC"2,3. Recently the chemistry and pharmacology of the thiophene analog 1-[1-(2-thienyl)cyclohexyl]-piperidine (TCP) have been described4, and within the last two years this compound has also appeared in the illicit drug trade3,5,6.
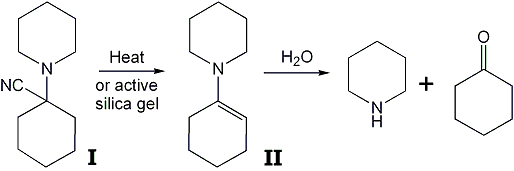
Imperfectly synthesized PCP has been reported to lead to distressful complications including coma and death7, although the nature of the impurities was not reported. The usual preparation of both PCP and TCP employs the intermediate 1-piperidinocyclohexanecarbonitrile (PCC; I)4,8,9 which can be removed from the final product only with difficulty. The presence of variable amounts of this contaminant has been correlated with undue toxicologic side-effects10, and there is a need for an analytical procedure which would allow its detection. This compound is unstable both to gas-liquid (GLC) and thin-layer chromatographic (TLC) methods in that through the abstraction of HCN the enamine II is formed, which in turn degrades to the components cyclohexanone and piperidine. When small amounts of PCC are subjected to GLC analysis only the product II is observed, whereas with the injection of larger quantities unchanged PCC is also apparent. A transition population between these two peaks indicates continuous transformation. With the usual TLC techniques the nitrile (PCC) is degraded to a variable extent by the act of being applied in solution to an activated plate. In this latter case, the generated enamine (II) is further degraded during actual chromatography.
We wish to describe an analytical procedure that minimizes the degradation of PCC as well as allowing separation and characterization of mixtures of PCP and TCP.
Materials And Methods
PCP and TCP were obtained as their hydrochloride salts from Parke Davis (Detroit, Mich., U.S.A.). Reference samples of PCC and of piperidinocyclohexene (II) (in benzene solution) were prepared as described8,9.
Analytical GLC analyses were performed on a Varian 1400 equipped with a flame ionization detector, employing a 6' x 1/8" I.D. glass column with 3 OV-17 on Chromosorb W at 158°C. Preparative GLC separations employed a Varian 90-P equipped with a thermal conductivity detector, with a 10' x 3/8" O.D. aluminum column with 10% OV-17 on Chromosorb W at 190°C.
TLC analyses employed Merck 0.25-mm silica gel 60 prepared glass plates, and the solvent system benzene-acetone-pyridine (16:8:1). Visualization reagents were ninhydrin (0.8% in acetone) and iodoplatinate (4 g chloroplatinic acid and 24 g KI in 1 L water, diluted after standing with 1 L methanol).
Infrared spectra were obtained on a Beckman IR-18 grating spectrophotometer.
Results And Discussion
Gas-liquid chromatographic analysis
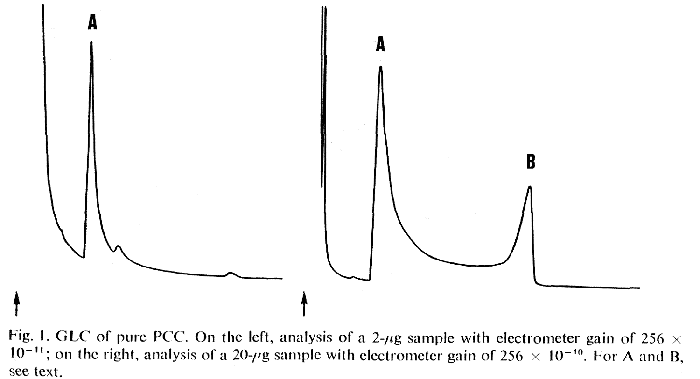
Typical chromatograms of a pure sample of PCC are shown in Fig. 1. The faster peak A is the major peak seen when small amounts of PCC are injected (1-2 ug). Its retention time corresponds to that of reference enamine II. A sample obtained from a preparative GLC run (with appropriate protection from the atmosphere) provided an infrared spectrum identical to that of synthetic II. A small peak immediately following A is absent in reference II. A tentative structural assignment of 1-cyclohexyl-1,2,3,4-tetrahydropyridine (an enamine isomeric with II) is supported by the generation of this minor component from II following treatment with ethanol and thermal cracking.
The second chromatogram shown in Fig. 1 resulted from the injection of 10 times as much PCC (20 ug) and was recorded at one-tenth the amplification. Peak B presumably represents unchanged PCC and the area above the projected baseline between A and B suggests the generation of A from the slower-moving B during passage through the column.
Thin-layer chromatographic analysis
If one employs usual TLC techniques (plate activation with heat followed by storage in a dry atmosphere), the chromatographic development of PCC results in the generation of two ninhydrin-positive spots in inconsistent proportions. The faster-moving component was undecomposed nitrile (PCC) and the second component, remaining at the origin in all but highly polar or highly basic solvent systems, proved to be piperidine. In experiments where PCC was applied to a warmed TLC plate using direct heat for solvent evaporation, only the second spot was observed indicating complete decomposition at the site of application. Attempts to chromatograph the enamine II likewise provided only piperidine as a developed spot.
Analytical procedure obviating decomposition of PCC
By using an unactivated TLC plate (open to ambient room air conditions), avoiding any heat during the application of the sample, and pre-wetting the site of application prior to spotting, the decomposition of PCC was largely circumvented.
A small drop of the chromatographic development solvent mixture is placed at the origin and allowed to evaporate. As soon as the apparent dampness has disappeared, but before the residual discoloration is also lost (the spot is still chalky white against a dull white background), a methanolic extract of the suspect drug is applied with a microcapillary. After air-drying without heat, the chromatogram is developed for about 10 cm, air-dried until largely free of pyridine smell, and then heated in an air oven at 110°C for 10 min. While still hot, it is sprayed with ninhydrin. The plate is allowed to cool, and oversprayed with iodoplatinate. The color developments and Rf values of the various materials assayed are given in Table 1.
| Compound | Rf | Color with ninhydrin | Color with iodoplatinate |
| Piperidine | 0.00 | purple | unchanged, fading to a bleached spot on standing. |
| PCP | 0.24 | no color | grey, permanent. |
| TCP | 0.49 | purple | unchanged, fading to a bleached spot on standing. |
| PCC | 0.70 | purple | unchanged, fading to a bleached spot on standing. |