Codeine to Morphine Conversion Reviewby Rhodium IntroductionThis text deals with four known methods of converting codeine to morphine by demethylation (codeine is morphine 3-methyl ether). Cleavage of aromatic ethers are commonly effected by reflux with concentrated HBr or HI. This relatively simple method can unfortunately not be used on codeine, as the oxygen bridge at the 9,10 position on the morphinan carbon skeleton would also rupture, causing the rearrangement of the molecule to the very potent emetic apomorphine, completely devoid of opiate-like effects. The author takes no responsibility whatsoever of whatever the reader might do with the information contained in this document. Keep in mind that the procedures described herein are probably illegal to carry out in all civilized parts of the world. Suggestions of alternative demethylation methods or anecdotal reports of the use of the syntheses below are welcomed. 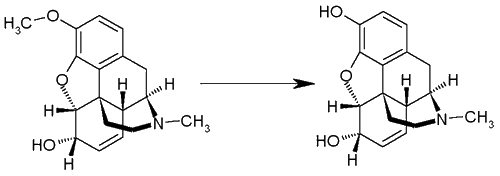
Figure 1: The Demethylation of Codeine to Morphine Using Pyridine HCl [1,2]A mixture of 1.00 g. of codeine and 3 g. of pyridine hydrochloride was placed in a bath at 220°C and heated for six minutes in a nitrogen atmosphere, after which the reaction mixture was immediately cooled and dissolved in 20 ml. of water, basified with 10 ml. of 4 N sodium hydroxide, and the non-phenolic material was removed by extraction with four 15-ml. portions of chloroform. The combined chloroform extracts were washed with 10 ml. of 0.5 N sodium hydroxide and 10 ml. of water, and the aqueous phase, after adding the washings, was adjusted to pH 9 and cooled thoroughly to precipitate phenolic material. After filtering and drying, this phenolic material was digested with 75 ml. of methanol, the mixture was filtered hot and the filtrate was chromatographed on an alumina (Merck and Co., Inc.) column (120x11 mm) using 700 ml. of methanol as eluent. The residue after evaporation of the methanol was dissolved in 10 ml. of 0.2 N sodium hydroxide, filtered, and the filtrate was adjusted to pH 9, precipitating the crude morphine. After drying, this crude morphine was sublimed (180-190°C (0.1 mm)), and the sublimate was crystallized from absolute ethanol. There was thus obtained a total of 210 mg. (22%) of morphine, m.p. 254-255°C. The "Homebake" Method [3]This is excerpted from a report dealing with clandestine manufacture of morphine and heroin from OTC codeine remedies, in so-called "homebake" laboratories in New Zealand. The method used is the same as the one introduced by Rapoport in 1951 [1], using pyridine hydrochloride. The authors report some perp's claims of 50% conversion from the codeine, but say they obtained 30% typically, and further state that this is about what one would expect from Rapoport's paper . Purity of up to 92% with a more typical purity in the 80% range was reported by the forensic chemists evaluating the method. The following is the actual procedure used in the clandestine labs, with some elaboration:
Report from someone using the above method Using Boron Tribromide [4]A solution of 2.99g (10 mmol) of anhydrous codeine in 25 ml of CHCl3 was added during 2 min to a well-stirred solution of 15g (59.9 mmol) of BBr3 in 175 ml of CHCl3 maintained in the range 23-26°C. A 10 ml portion of CHCl3, which was used to rinse the addition funnel, was added to the reaction mixture and stirring was continued for 15 min at 23-26°C. The reaction mixture was then poured into a well-stirred mixture of 80g ice and 20 ml of concentrated (28-30%) ammonia. The two-phase system was kept at -5°C to 0°C for 0.5h (continous stirring) and filtered. The resulting crystalline material was washed thorougly with small portions of cold CHCl3 and H2O and dried to give 2.67g (88.1%) of slightly off-white morphine hydrate, mp 252.5-254°C. Report from a chemist trying out the above method: I extracted the codeine using the NaOH/chloroform method. I then went through the synthesis - the BBr3 is really nasty - fuming toxic shit! I had a
high power exhaust fan in front of my work area and it took care of the fumes (all the insects outside the window died!). I dissolved the BBr3 in chloroform
and then added the codeine/chloroform mixture - lots of HBr gas evolved - stoppered the flask and lead the fumes into water, which dissolved the HBr gas.
The codeine made this funky pink precipitate. Continued to follow the steps, added mix to ammonia/ice (I had this in a alcohol/ice bath at -5°C). Once again
fumes evolved (not as much). Stirred 30 min. Filtered out a pale white-yellow gelatinous material, washed with cold chloroform and water and dried. It
seemed to have a bromine odor and was very acidic (I probably used a bit too much BBr3) so I washed with cold water again (morphine hydrate isn't water
soluble) and the pH normalized. I then dissolved it in dilute HCl, and neutralized to pH 6 with NaOH.
Using Sodium Propylmercaptide [5]A solution of 3.00 grams (10 mmol) of codeine in 60 ml of dry dimethylformamide was degassed under nitrogen by repeatedly stirring under vacuum, followed by inletting nitrogen. Following the addition of 3.00 grams (26.7 mmol) of potassium tert-butoxide, the degassing process was repeated, and 3.0 ml (32.7 mmol) of n-propanethiol was injected by syringe. The mixture was stirred at 125°C under nitrogen for 45 min (similar results at 110°C for 3h), cooled, and quenched with 3.0 ml of acetic acid. The solvent was removed under high vacuum, and the residue dissolved in 30 ml of 1N hydrochloric acid. The acid solution was washed with several portions of ether, treated with 5ml of 20% sodium bisulfite, and alkalized to pH 9 with ammonium hydroxide. The precipitated solid was collected, washed with water, and dried in vacuo (100°C) to leave 2.30g (80%) of morphine as tan crystals. Using L-Selectride (Lithium tri-sec-butyl Hydride) [6,7]482 mg (1.6 mmol) of codeine was dissolved in 4 ml of an 1 M solution of L-selectride in THF (4.0 mmol) and was refluxed for 3.5h. The reaction was quenched with water (5ml), followed by 2ml of 15% NaOH solution and removal of the THF. The resulting mixture was washed twice with CH2Cl2, cooled to 0-5°C and acidified to pH 1 with 10% HCl. After basification with ammonium hydroxide to pH 9, the mixture was extracted into CHCl3, the organic phase was washed with brine and dried over Na2SO4. Removal of the solvent, followed by recrystallization from water gave 355mg (73%) of morphine hydrate. Unreacted codeine was recovered from the non-phenolic extracts, and after purification by recrystallization from water, it amounted to 71mg (14%). References[1] H. Rapoport, The Preparation of Morphine-N-Methyl-C14, J. Am. Chem. Soc., 73, 5900 (1951)[2] H. Rapoport, Delta-7-Desoxymorphine, J. Am. Chem. Soc., 73, 5485 (1951) [3] K. Bedford, Illicit Preparation of Morphine from Codeine in NZ, Forensic Sci. Int. 34(3), 197-204 (1987) [4] K.C. Rice, A Rapid, High-Yield Conversion of Codeine to Morphine, J. Med. Chem., 20(1), 164 (1977) [5] J.A. Lawson, An Improved Method for O-Demethylation of Codeine, J. Med. Chem., 20(1), 165 (1977) [6] A. Coop, L-selectride for the O-Demethylation of Opium Alkaloids, J. Org. Chem., 63, 4392-96 (1998) [7] G. Majetisch, Hydride-Promoted Demethylation of Methyl Ethers, Tet. Lett. 35(47), 8727-8730 (1994) |