Synthesis of 4-allyl-pyrocatechol (3,4-dihydroxy-allylbenzene)[ Back to the Chemistry Archive ] Pyrocatechol mono-Allyl Ether (2-allyloxyphenol)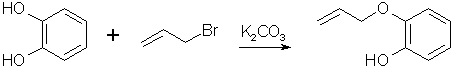
Pyrocatechol monoallyl ether [7] 110g (1 mole) catechol and 138g potassium carbonate (1 mole) was mixed in 200ml of acetone, with some evolution of heat. The flask containing this mixture was then fitted with a stirrer and attached to a reflux condenser. 121g (1 mole) of allylbromide was added slowly and with constant stirring. When about three-fourths of it had been added, external cooling of the flask was necessary to prevent too vigorous boiling. The remaining allylbromide was added with less evolution of heat. Refluxing on a water bath with continual stirring was continued for four hours. After cooling, the solid material was filtered off and washed with 50 mL of acetone. The washing were added to the first filtrate and the acetone was removed by distillation. The oil was then fractionally distilled under vacuum and the portion coming over at 108-113°C/18 mmHg was collected in a 70% yield. The allyl ether must be vacuum distilled under a good vacuum (at least 25 mmHg), or else it will rearrange. Pyrocatechol monoallyl ether [3] 100g Pyrocatechol (0.9 mol), 100g allyl chloride (1.3 mol), 15g NaI (0.1 mol) and 106g Na2CO3 (0.77 mol) was refluxed in anhydrous acetone to give a 75% yield of pyrocatechol monoallyl ether, bp 81°C/1.5 mmHg. Acetone is the preferred solvent for the reaction. In ethanol the reaction only gave approximately 45%, as did it in propanol (without the NaI), and in propanol with the NaI added the yield was 68%. 4-Allyl-Pyrocatechol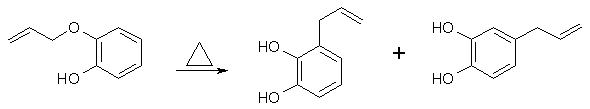
Base Catalyzed Claisen Rearrangement [1] The thermal rearrangement of catechol mono-allyl ether is reported [2,3,4] to give at 160-200°C a mixture of products, 3-allyl-catechol and 4-allylcatechol, in the ratio 5:4, apparently resulting from competing ortho- and para-Claisen rearrangements [5,6]. Our investigation of this reaction shows that the ratio of the two products and the rate of the rearrangement are sensitive to the pH at which the reaction is carried out. Thus in ethanolic HCl (2 drops HCl in 25 ml EtOH) at 180°C the ratio of 3-allyl- to 4-allylcatechol is 2:1 and in ethanol containing 1 equiv. of NaOEt at 78°C the reaction proceeds at approximately the same rate and the ratio 3-allyl- to 4-allylcatechol is 1:4. These preliminary observations suggested that the rearrangement of catechol mono-allyl ether is base catalysed and that the rearrangement of the anion of catechol mono-allyl ether takes a different course from that of the un-ionised phenol. Experimental: Equimolar amounts of the allyl ether/NaOEt, and then 2-3 times the weight of the ether in ANHYDROUS ethanol as solvent. Reflux for a few hours and then evaporate the solvent, and take up the residue in 2M NaOH. Wash with DCM, acidify and extract with DCM. Evaporate the solvent and fractionately distill the residue to separate the allyl-catechols. Allylpyrocatechols [3] When 100g Pyrocatechol mono-Allyl Ether was heated 35-40 min at 160-170°C an exothermal reaction with a temperature jump to 270°C took place, producing a liquid which on fractional distillation, gave 35-38% 3,4-dihydroxy-1-allylbenzene (4-allylcatechol) bp 123-5°C/1-2mmHg, mp 48°C, and 47.5% 2,3-dihydroxy-1-allylbenzene (3-allylcatechol) bp @ 95°C/5mmHg, mp 24°C. 4-Allylcatechol [8] To a solution of 11.5 g (0.07 mol) of eugenol in 30 mL of dry THF was added stepwise 2.1 g (0.07 mol) of a dispersion of NaH in oil (80%). After the evolution of gas had stopped a solution of 13.4 g (0.07 mol) of LiPPh2 in 60 mL of THF was added. After stirring for 3 h, 2 mL of water was added and the mixture was poured into 200 mL of degassed 0.2 N aqueous NaOH. The yellow solution was washed with ether (4x). and the combined organic layers were extracted with 100 mL of 0.2 N aqueous NaOH. The combined water layers were acidified with concentrated HCl to pH=6. The resulting emulsion was extracted with ether, and the organic layers were washed with water (5x), dried (MgSO4), and evaporated to dryness. Purification by column chromatography (eluent: 2% MeOH in CHCl3) gave 5.97 g (57%) of a brownish oil which could be further purified by sublimation. A yield of 3.9 g (37%) of 4-allylcatechol was obtained as a bright white solid. 4-Allylcatechol [9] A solution of 354 ml (2.30 mmol) of eugenol and 292 g (6.89 mol) of lithium chloride in 3.7 L of N,N-dimethylformamide was refluxed for a total of 44 hours (h), and after 4 h, 18 h and then 7 h, a further 292 g (6.89 mol) of lithium chloride were added each time. After cooling, 2 L of toluene were added and the resultant precipitate was filtered off with suction and extracted with toluene. The organic extracts were combined and concentrated on a rotary evaporator. After flash chromatography (ether/pentane, 1:1, Rf = 0.37) on silica gel, 173 g (50%) of 4-allylcatechol were obtained. Physical properties of 4-allylcatechol Alternate names: 3,4-dihydroxy-allylbenzene, 4-allylpyrocatechol. Melting point: 46-48.5°C Boiling point: 123-124°C/1mmHg, 141-144°C/7mmHg, 147-149°C/10mmHg, 155-157°C/13mmHg, 156-158°C/16mmHg. Solubility (H2O): 25 g/l at 26°C. Recrystallization solvents: Benzene, petroeleum ether, diethyl ether. Cognate Syntheses: Allylation of guaiacol (2-methoxyphenol) with allyl bromide, followed by Claisen rearrangement of the formed 2-allyloxyanisole gives 2-hydroxy-3-methoxyallylbenzene (o-Eugenol) in good yield [10]. This can in turn be used as a precursor for asarone and other interesting compounds. References [1] J. Chem. Soc. Chem. Comm. 494 (1974)
|