Synthesis of Allyl Alcohol[ Back to the Chemistry Archive ] ProcedureA mixture of 500g anhydrous oxalic acid and 500g of glycerol was heated in a partial vacuum on a water bath for 4-5h (or longer) until formic acid ceased to distill over. The mixture was then gradually heated to 240°C under ordinary pressure, the flask being fitted with a fractionating column. At 220-225°C CO2 was given off and a mixture of approximately equal amounts of allyl alcohol and allyl formate distilled over leaving in the distillation flask a residue containing somewhat 50% of the glycerol originally used. Practically no acrolein was produced. The distillate was treated with 50g NaOH in 1000ml water (to hydrolyze the formate), allowed to stand for 12h at room temp, and finally distilled. The first 300 ml of distillate contained all the allyl alcohol, which after fractionation yielded 200-210g of a allyl alcohol/water mixture (bp 87-88°C) which may be dehydrated using anhydrous potassium carbonate yielding approximately 150g of anhydrous allyl alcohol. The glycerol residue may be made up to 500g with fresh glycerol, and the procedure repeated. After 4-5 repetitions the residue should be discarded though, as the tendency to froth during the distillation increases. The yield of allyl alcohol is nearly quantitative calculated on the amount of glycerol reacted. Note that formic acid is produced in the reaction. This can be used for many purposes, see the Formic Acid Preparation doc. Reference: J. Chem. Soc. Vol 107, p407-10 (1915) Allyl Alcohol from Glycerol and Formic Acid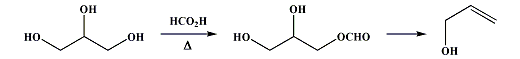
Instead of oxalic acid, formic acid can advantageously be used instead, as detailed in Organic Syntheses. |