A new route to
2,6-dimethoxy-4-methylamphetamine (2,4,6-DOM)
by the Labrat
Introduction
This is a very quick route to make the 2,4,6-isomer of DOM, probably as active as the parent compound and opens up a new route
to make more 4-alkyl substituted compounds. The 2,4,6-isomers of DOET and DOPR can be synthesized by substituting
3,4,5-trimethoxytoluene with 3,4,5-trimethoxyethylbenzene and 3,4,5-trimethoxypropylbenzene respectively. 3,4,5-trimethoxytoluene
can be synthesized from the highly useful 3,4,5-trimethoxybenzaldehyde by Clemmensen reduction as described in Organic Synthesis
Collective IV: 204 ('63) for preparation of creosol:
3,4,5-trimethoxytoluene from 3,4,5-trimethoxybenzaldehyde
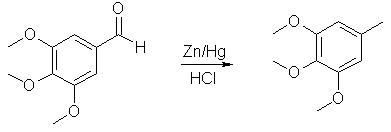
In a 5 l flask is placed 1.5 kg of amalgamated zinc and 800 ml of concentrated HCl. The flask is heated to cause gentle
refluxing to occur. A solution of 150 g trimethoxybenzaldehyde in 450 ml of 95% ethanol and 1.5 l of concentrated hydrochloric
acid is added dropwise through the dropping funnel over an 8-hr period. After the addition the mixture is refluxed for 30 minutes
more. The liquid, consisting of two layers, is decanted from the zinc (this can be recycled by adding more zinc and renewing
amalgamation). The aqueous layer is removed and washed with 3x200 ml benzene. The extracts are combined with the crude creosol.
The residual amalgamated zinc is washed with 2x100 ml benzene, add this to the pooled extracts. The extracts are washed with
2x200 ml 5% bicarbonate solution and the resultant precipitate of metallic salts is removed by filtration. The solution is washed
with 200 ml water. The benzene is removed under pressure. The residue is distilled to give a ~65% yield of 3,4,5-trimethoxytoluene.
The next step in the synthesis is a regioselective reductive electrophilic substitution on the 3,4,5-trimethoxytoluene as is
done in J. Org. Chem. 55, 5387-90 (1990) and Tet. Lett. 30, 1689-90 ('89). WHAT is the Labrat saying? Well, actually its quite simple. The
2-methoxygroup is reduced by sodium in THF creating an aryl-anion, stabilised by the two ortho methoxy group. This reaction is
pretty new, but comparable to lithiation reactions or Birch reduction (in aprotic solvents). Then, the anion is quenched with an
electrophilic substance to get a 2-substituted compound. There are a lot of electrophiles possible, but to make the 2,4,6-DOM,
Labrat's choice was 1,2-dibromopropane. This electrophile was chosen, because the aryl anion will only react with PRIMARY, not
secondary halides. Here are experimental details:
1-(2,6-dimethoxy-4-methylphenyl)-2-Br-propane from 3,4,5-trimethoxytoluene
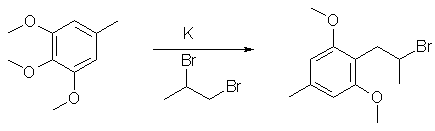
A solution of 5,1 (28 mmol) of 3,4,5-trimethoxytoluene in 5 ml anhydrous THF was added dropwise to a mixture of 3,3 g (0.084 g
atom) freshly cut K metal in 20 ml anhydrous THF and vigorously stirred at room temperature. The mixture was stirred for 24 hrs at
room temperature and then chilled to 0°C. Then 8,5 g (42 mmol, 1.5 eq) 1,2-dibromopropane dissolved in 5 ml anhydrous THF is
added slowly and the resulting mixture was stirred for 4 hrs. The reaction was quenched by dropwise addition of 10 ml H20 and
extracted with 3x30 ml Et2O. The organic phase was collected, washed with 50 ml H2O, dried with CaCl2 and evaporated to afford a
70-80% yield of crude product, which can be purified by distillation or recrystallisation.
This is a quick and relatively easy process. There are however a few things to remember:
- Using Na instead of K gave complex results
- A side reaction is reductive demethylation, producing phenols which should be saved (they can be extracted with acid).
Those phenols can be realkylated again, this time maybe with diethyl sulfate to produce Tweetios.
- As the alkylchain on the 1-position gets longer, the yield of the aryl anion gets smaller and consequently so is the total yield.
There are a lot of ways to produce amphetamines from the isopropyl bromide. Ammonolysis is just one way, but the yield is low
at low pressure. A much better way is presented in Synthesis 202 ('85). The article describes a variant of the Staudinger-reaction:
the phosphite-azide coupling method. This reaction uses more common and cheaper chemicals then the original Staudinger reaction.
This synthesis is essentially three steps in a one-pot procedure:
- Azidation of the secondary bromide
- The Staudinger reaction of the azide with triethyl phosphite
- Two-step deprotection of the iminophosphorane by means of HCl at room temp.
2,4,6-DOM from 1-(2,6-dimethoxy-4-methylphenyl)-2-Br-propane
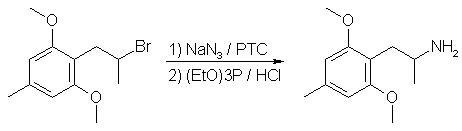
A mixture of 6.5 g (0.1 mol) NaN3, 0.05 mol of the bromide, 5% tetrabutylammonium bromide and 30 ml benzene is refluxed with
efficient stirring for 12 hrs. The product is cooled to room temp and filtered by suction. The precipitate of inorganic
salts is washed with 2x10ml benzene. The filtrate is washed with 2x20ml water and dried with MgSO4. Triethyl phosphite (8.3g,
0.05 mol) is added dropwise with stirring to the solution of the organic azide thus obtained. The temperature of the slightly
exothermic solution is kept below 30°C by occasional cooling. The solution is left overnight at room temp. It is then
satured with dry HCl gas for 2h at room temperature and again allowed to stand overnight. The mixture is evaporated under
reduced pressure. Dry ether (30 ml) is added to the residue and the mixture is refrigerated for 24 hrs. The precipitated amine
hydrochloride is isolated by suction, washed with 2x15 ml ether and dried in vacuo over NaOH. Yield ~70%.
I accept no responsibility for the misuse of this information. Lr
|